Description
Healgen 9-in-1 Respiratory Combo Antigen Test
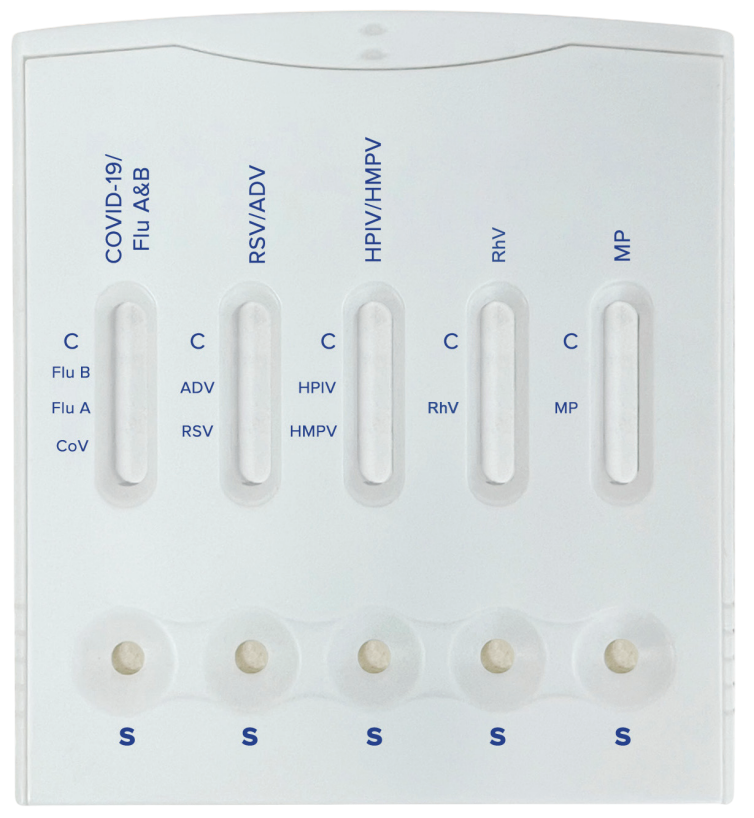
The Healgen 9-in-1 Combo Antigen Test is a comprehensive rapid test designed for the qualitative detection of multiple common respiratory pathogens from a single specimen. It targets SARS-CoV-2 (COVID-19), Influenza A, Influenza B, Respiratory Syncytial Virus (RSV), Adenovirus, Human Parainfluenza Viruses (HPIV), Human Metapneumovirus (HMPV), Rhinovirus (RhV), and Mycoplasma pneumoniae (M. pneumoniae).
Why Choose a 9-in-1 Panel?
- Broad coverage: Helps differentiate leading causes of acute respiratory infection in one workflow.
- Fast turnaround: Visual results in approximately 15 minutes (read within the time window stated in the IFU).
- Streamlined workflow: Single collection, minimal handling, and no instrumentation required.
- Point-of-care friendly: Room temperature storage (2–30°C / 36–86°F) supports flexibility across clinical settings.
Intended Use
For the qualitative detection of specific antigens from the pathogens listed above in human respiratory specimens. For professional in vitro diagnostic use where permitted by local regulations. Results should be interpreted together with clinical findings and, when indicated, confirmed by additional laboratory methods.
Specimen & Handling
- Specimen types: Anterior nasal swab and/or nasopharyngeal (NP) swab (refer to the IFU for accepted matrices).
- Storage: 2–30°C (36–86°F). Do not freeze. Keep away from direct sunlight and humidity.
- Time to result: ~15 minutes (do not read after the maximum read time in the IFU).
- Shelf life: Up to 24 months from the date of manufacture (see kit label for expiry).
Kit Components
- Test cassettes (individually pouched)
- Extraction buffer/tubes and caps
- Sterile swabs
- Package insert / Instructions for Use (IFU)
Key Benefits
- Detects nine targets in one panel: COVID-19, Flu A, Flu B, RSV, Adenovirus, HPIV, HMPV, Rhinovirus, and M. pneumoniae.
- Rapid visual readout with no specialized equipment.
- Supports triage during peak respiratory seasons and outbreak investigations.
- Compact kit; easy to store and deploy at the point of care.
Important Notes
- Negative results are presumptive and do not rule out infection; test again if clinically indicated.
- Positive results do not exclude co-infections with other organisms.
- Follow local guidelines for confirmatory testing and reporting.
Pathogen Coverage
| Target | Pathogen |
|---|---|
| 1 | SARS-CoV-2 (COVID-19) |
| 2 | Influenza A |
| 3 | Influenza B |
| 4 | Respiratory Syncytial Virus (RSV) |
| 5 | Adenovirus |
| 6 | Human Parainfluenza Viruses (HPIV) |
| 7 | Human Metapneumovirus (HMPV) |
| 8 | Rhinovirus (RhV) |
| 9 | Mycoplasma pneumoniae |
Specifications
| Specification | Details |
|---|---|
| Assay Principle | Immunochromatographic (lateral flow) assay for qualitative antigen detection |
| Specimen | Anterior nasal swab / Nasopharyngeal swab (see IFU) |
| Time to Results | ~15 minutes |
| Operating Conditions | 2–30°C (36–86°F), dry environment |
| Shelf Life | Up to 24 months (refer to packaging) |
| Use | Professional use only, where permitted |
Ordering Information
| Product Description | Specimen | Catalog No. | Format | Kit Size | Qualification |
|---|---|---|---|---|---|
| Healgen 9-in-1 Respiratory Combo Antigen Test | Nasal / Nasopharyngeal swab | (On request) | Cassette | 20 Tests/Kit | CE/Local market status: see IFU & label |